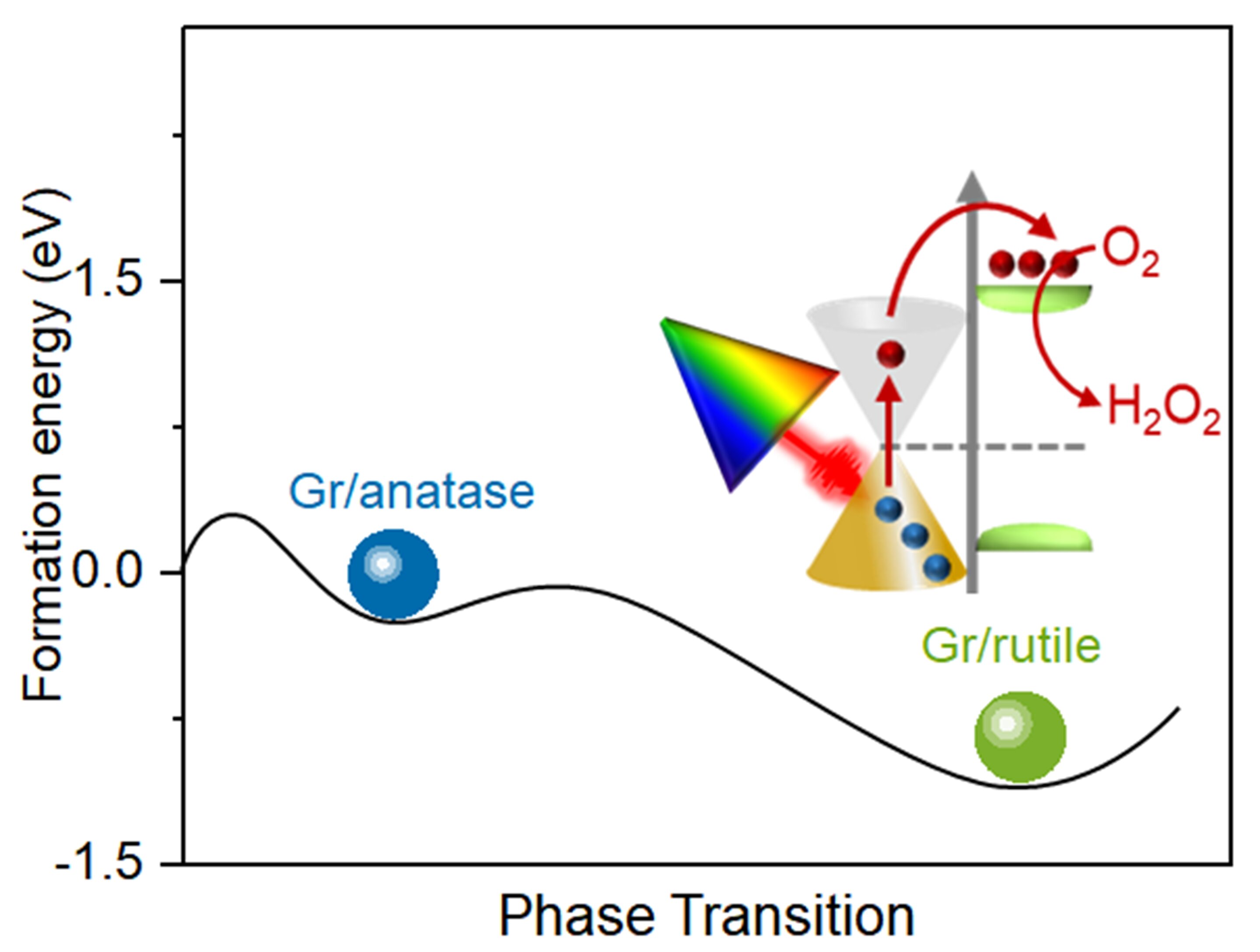
Improve hydrogen peroxide production through sustained photocatalysis
The 'Substance identity' section is calculated from substance identification information from all ECHA databases. The substance identifiers displayed in the InfoCard are the best available substance name, EC number, CAS number and/or the molecular and structural formulas. Some substance identifiers may have been claimed confidential, or may.

16 Insanely Useful Hydrogen Peroxide Cleaner Recipes
Hydrogen peroxide naturally occurs in the body as a byproduct of cellular respiration. As a chemical, it is used in bleaching, cleansing, and disinfecting processes in households and industries. Household concentrations generally range from 3 to 9% and are most commonly found at 3%. Industrial concentrations can be as high as 70%, most commonly ranging between 27.5 to 70%.[1]

Een flesje waterstofperoxide kost 0,99 cent bij de Kruidvat. Moet je zien wat je ermee kan!
2. Metabolism. Hydrogen peroxide H 2 O 2 is a non-radical reactive oxygen species (ROS) and it, like singlet oxygen 1 O 2 and free radicals such as superoxide anion O 2 − and hydroxyl radical • OH, is one of the major members of the ROS family [].In contrast to other ROS, H 2 O 2 is relatively stable, with a half-life of ms and its level in a plant leaf oscillates around 1 µmol per gram.

מי חמצן 17.5 להידרופוניקה Hydrogen Peroxide Naturetech Shop
There's no scientific evidence that drinking hydrogen peroxide yields benefits. Plus, doing so is linked to dangerous side effects, including breathing problems, severe gut damage, and death.

HYDROGEN PEROXIDE 100ML BB
The most dangerous side effects of drinking hydrogen peroxide can be simplified into three groups. Direct caustic injury. When too much hydrogen peroxide is consumed, internal tissue in the throat.

Buy Hydrogen Peroxide 32 Solution, Lab Grade 33+ Bulk Sizes
In microdroplets of water, just millionths of a meter wide, a portion of the H 2 O molecules present can convert into a close chemical cousin, hydrogen peroxide, H 2 O 2, a harsh chemical commonly.

Hydrogen Peroxide 34 32oz Shop Renowned Chemicals
Hydrogen peroxide is a colorless liquid at room temperature with a bitter taste. Small amounts of gaseous hydrogen peroxide occur naturally in the air. Hydrogen peroxide is unstable, decomposing readily to oxygen and water with release of heat. Although nonflammable, it is a powerful oxidizing agent that can cause spontaneous combustion when it.

Hydrogen Peroxide 3 BP Food Grade (10 vols) APC Pure
Hydrogen peroxide (H 2 at 20°C) that has strong oxidizing properties. It is commonly used (in concentrations typically around 5%) to bleach human hair, hence the phrases "peroxide blonde" and "bottle blonde". It burns the skin upon contact in sufficient concentration. In lower concentrations (3%), it is used medically for cleaning wounds and.

Buy Care Hydrogen Peroxide Solution 9 200ml Online
A 2009 study found that rinsing in hydrogen peroxide reduced the bacterial count by 85 percent. 20. Do use it to sterilize makeup brushes. After washing excess makeup off your brushes with a.

Hydrogen peroxide Wikipedia
What is hydrogen peroxide? Hydrogen peroxide is water (H2O) with an extra oxygen molecule (H2O2). "The extra oxygen molecule oxidizes, which is how peroxide gets its power," says Dr. Beers. "This oxidation kills germs and bleaches color from porous surfaces like fabrics." When you use peroxide, go for medical-grade peroxide, which is 3% strength.

5 Natural Ways To Treat Vaginal Odour
The hydrogen peroxide plasma decomposes into water and oxygen and the chamber is brought back to atmospheric pressure. Hydrogen peroxide, often mixed with <50 ppm Ag cations or peracetic acid, is emitted through a nozzle, producing an aerosol of approximately 8-10 micrometer particles which can interact and decontaminate items in the treatment.

Hydrogen Peroxide H2O2 Oxy Già Công Nghiệp 50
Hydrogen peroxide is a chemical compound with the formula H 2 O 2.In its pure form, it is a very pale blue liquid that is slightly more viscous than water.It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%-6% by weight) in water for consumer use, and in higher concentrations for industrial use.Concentrated hydrogen peroxide, or "high-test peroxide.

Hydrogen peroxide kopen kruidvat
The global utilization of hydrogen peroxide, a green oxidant that decomposes in water and oxygen, has gone from 0.5 million tonnes per year three decades ago to 4.5 million tonnes per year in 2014, and is still climbing. With the aim of expanding the utilization of this eminent green chemical across different industrial and civil sectors, the.
/s3/static.nrc.nl/images/gn4/stripped/data56317635-2b877d.jpg)
Hydrogen Peroxide Kruidvat ZHH85 AGBC
Hydrogen peroxide is an ingredient in many bleaches, dyes, cleansers, antiseptics, and disinfectants. It has many potential uses, such as for skin care and oral hygiene. However, there are also.

20 Different Ways To Use Hydrogen Peroxide
Decomposition. Hydrogen peroxide is thermodynamically unstable and decomposes to form water and oxygen with a ΔH ⦵ of -98.2 kJmol-1 and a ΔS ⦵ of 70.5 Jmol-1 K-1.. 2 H 2 O 2 → 2 H 2 O + O 2 The rate of decomposition increases with rising temperature, concentration and pH, with cool, dilute, acidic solutions showing the best stability. Decomposition is catalysed by various compounds.

IV Treatments
Hydrogen peroxide is produced physiologically by oral bacteria and plays a significant role in the balance of oral microecology since it is an important antimicrobial agent. In the epithelial cells, the enzyme superoxide dismutase catalyzes a reaction leading from hydrogen peroxide to the ion superoxide. The induced oxidative stress stimulates.